All nascent polypeptides pass through a tunnel in the large ribosomal subunit before they are released into the cytosol or into the lumen of the ER. Upon arrival at the ribosomal tunnel exit nascent polypeptides encounter a group of proteins, which mediate further steps of protein biogenesis and is therefore termed ribosome-associated protein biogenesis factors (RPBs). Depending on their properties, different nascent polypeptides require different RPBs.
We are interested in the function and mechanism of RPBs in eukaryotic cells with a focus on the yeast Saccharomyces cerevisiae.

This is the 60S large ribosomal subunit displaying the ribosomal exit tunnel. A nascent chain, which is still bound to the ribosome as a peptidyl-tRNA, is depicted inside of the tunnel. The peptidyl transferase center (PTC) is indicated. Three of the almost 50 large subunit ribosomal proteins are highlighted: Rpl17 (uL22), Rpl4 (uL4), and Rpl39 (eL39). These ribosomal proteins expose domains within the lumen of the ribosomal tunnel, which are important for the early steps of protein folding and targeting.
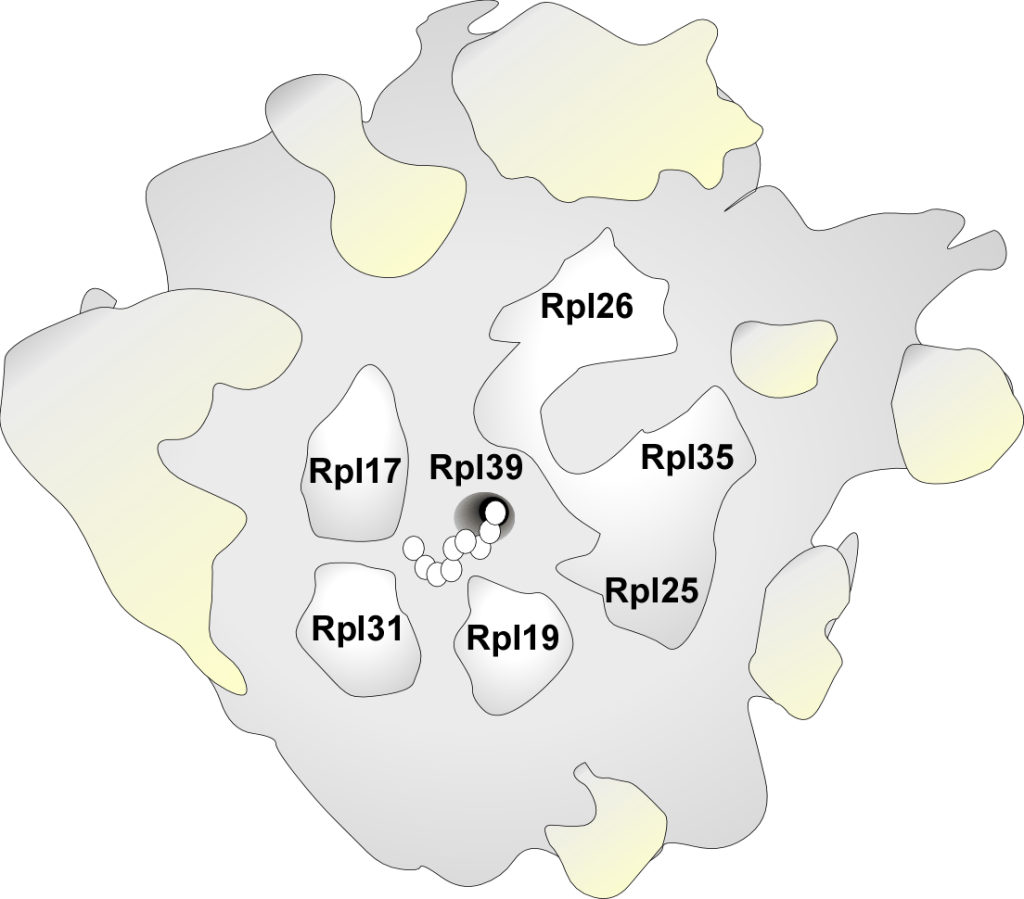
RPBs often interact with ribosomal proteins, which form a platform surrounding the tunnel exit. At this position, RPBs can easily contact nascent chains when they emerge from the tunnel exit.
There is a whole zoo of RPBs, which perform diverse functions – we are interested in all of them! However, we focus on those RPBs, which belong to the groups of chaperones and targeting factors.
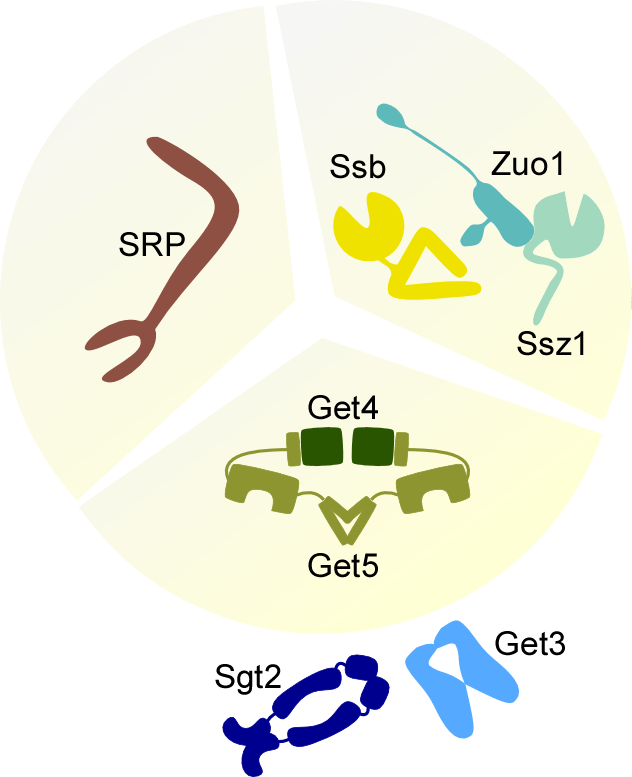
- Ssb is a chaperone of the Hsp70 family. Ssb binds to the ribosome and also to nascent chains. The ribosome-associated complex (RAC) consists of an Hsp40 homolog termed Zuo1 and an Hsp70 homolog termed Ssz1. RAC binds directly to the ribosome and acts as a cochaperone of Ssb. Further reading: (Zhang et al, 2017) The RAC/Ssb chaperone system does not only facilitate the folding of newly synthesized polypeptides, but is also involved in the regulation of metabolic signaling. Further reading: (Hübscher et al, 2017)
- Signal recognition particle (SRP) is a large multisubunit complex composed of several proteins and an RNA. SRP binds directly to the ribosome and, via a subunit termed Srp54, to nascent chains containing an ER targeting signal. Binding of nascent chains to SRP results in cotranslational targeting to the endoplasmic reticulum. Further reading (Akopian et al, 2013)
- The Get4/5 complex is a component of the guided entry of tail-anchored proteins (GET) pathway. The GET pathway assists the posttranslational delivery of tail-anchored proteins containing a single C-terminal transmembrane domain to the endoplasmic reticulum. Further reading (Borgese et al, 2019). Even though the GET pathway takes place posttranslationally, Get4/5 binds directly to ribosomes (Zhang et al. 2016). We recently found that ribosome-bound Get4/5 recruits the chaperone Sgt2, which binds and shields hydrophobic tail-anchor sequences (Zhang et al, 2021).
You must be logged in to post a comment.